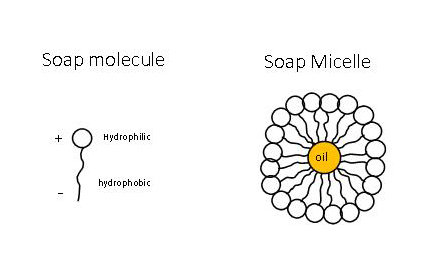
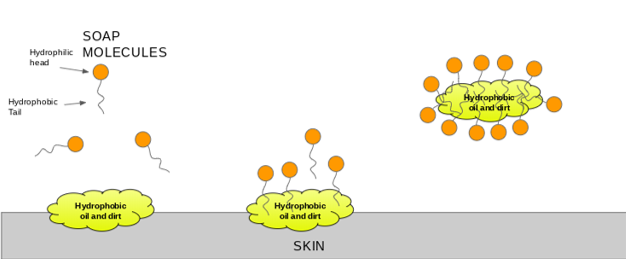
Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents like ethanol also?
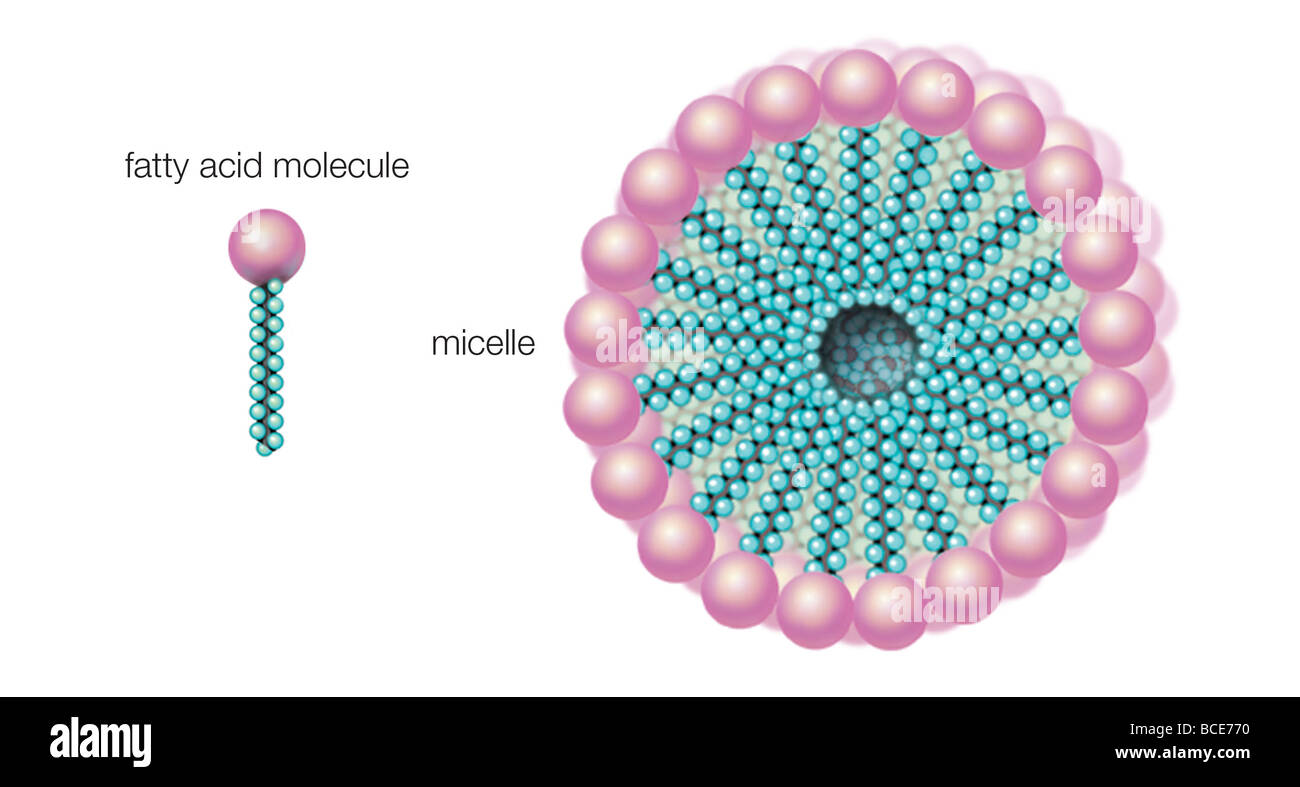
When dissolved in water, fatty acids in soap form micelles, the hydrophilic ends turned outward and the hydrophobic ends inward Stock Photo - Alamy

Chemistry Quizzes - • How soaps clean:- • Soaps are sodium or potassium fatty acids salts, produced from the hydrolysis of fats in a chemical reaction called saponification. • Each soap molecule


![MCQ] In the soap micelles (a) the ionic end of soap is on the surface MCQ] In the soap micelles (a) the ionic end of soap is on the surface](https://d1avenlh0i1xmr.cloudfront.net/aed2074f-aafe-447a-b1f0-adb685bf01d5/structure-of-a-soap-micelle---teachoo.jpg)








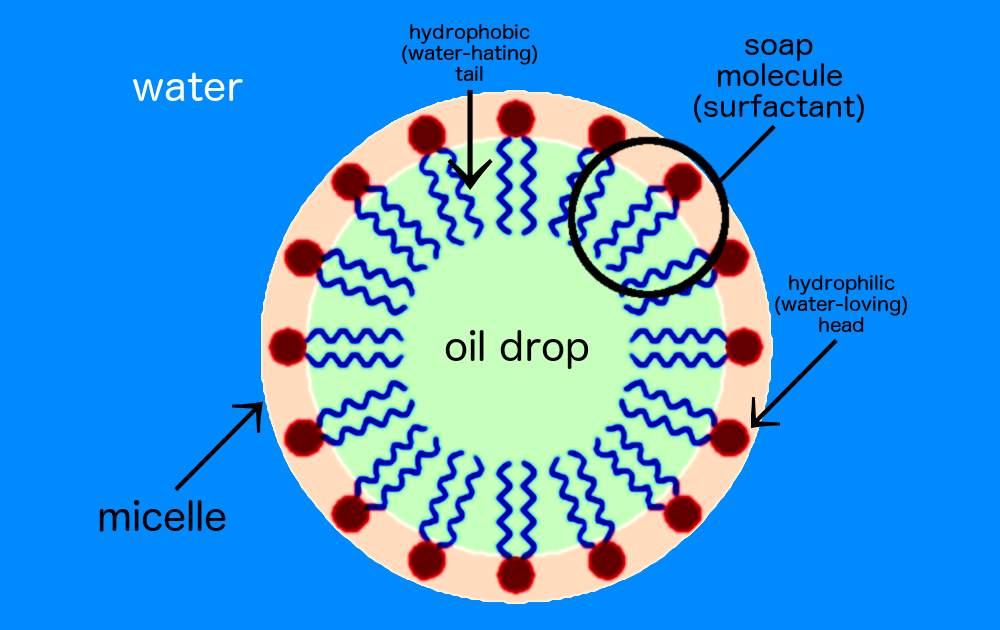


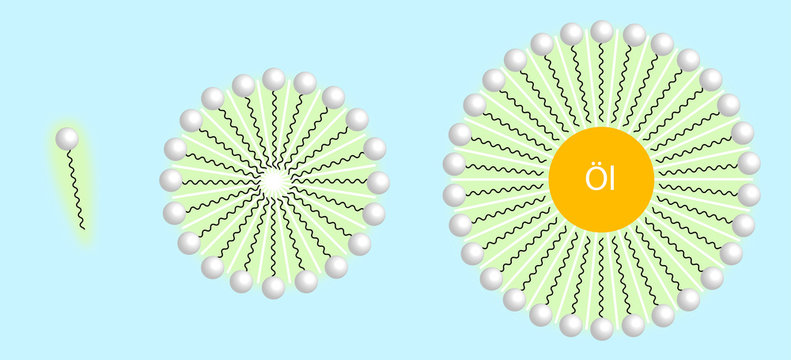
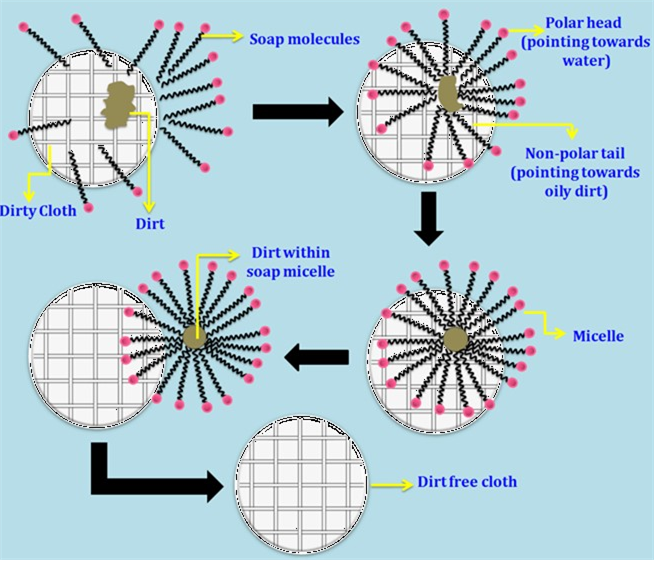
![Class 10 Carbon and its compounds] What are Micelles? - Teachoo Class 10 Carbon and its compounds] What are Micelles? - Teachoo](https://d1avenlh0i1xmr.cloudfront.net/c5536fd4-cc1f-4684-a708-ece69fada9ca/structure-of-micelle---teachoo.jpg)