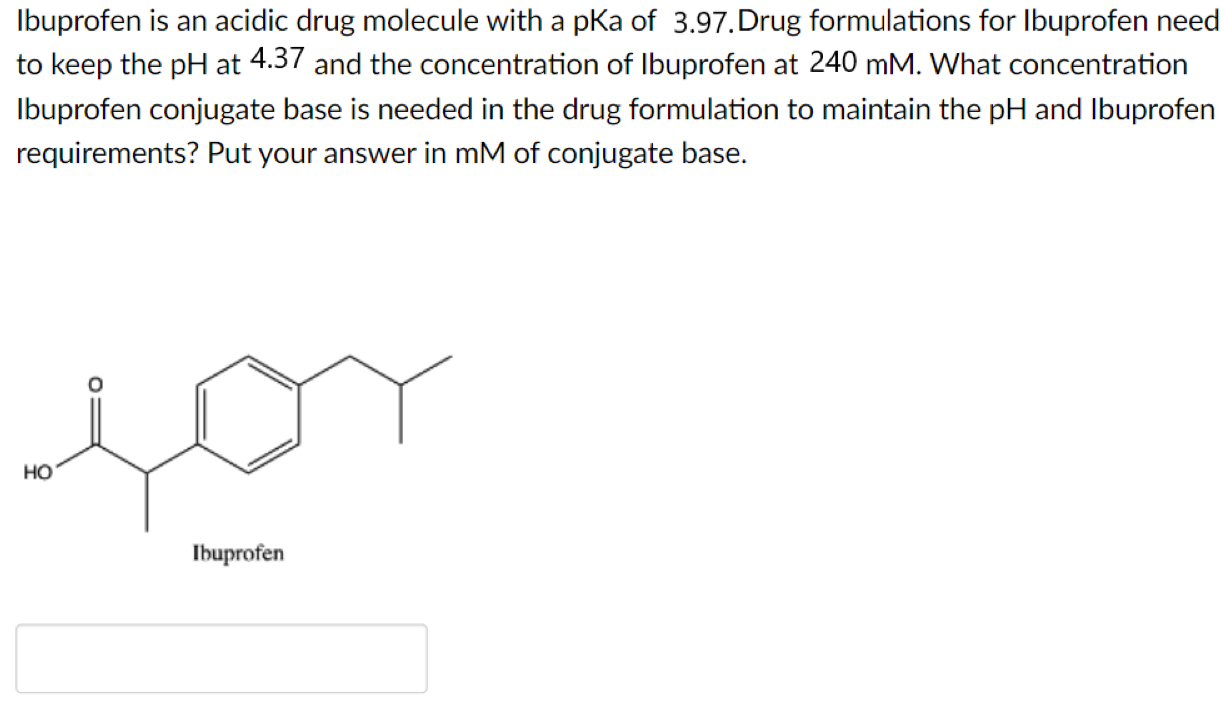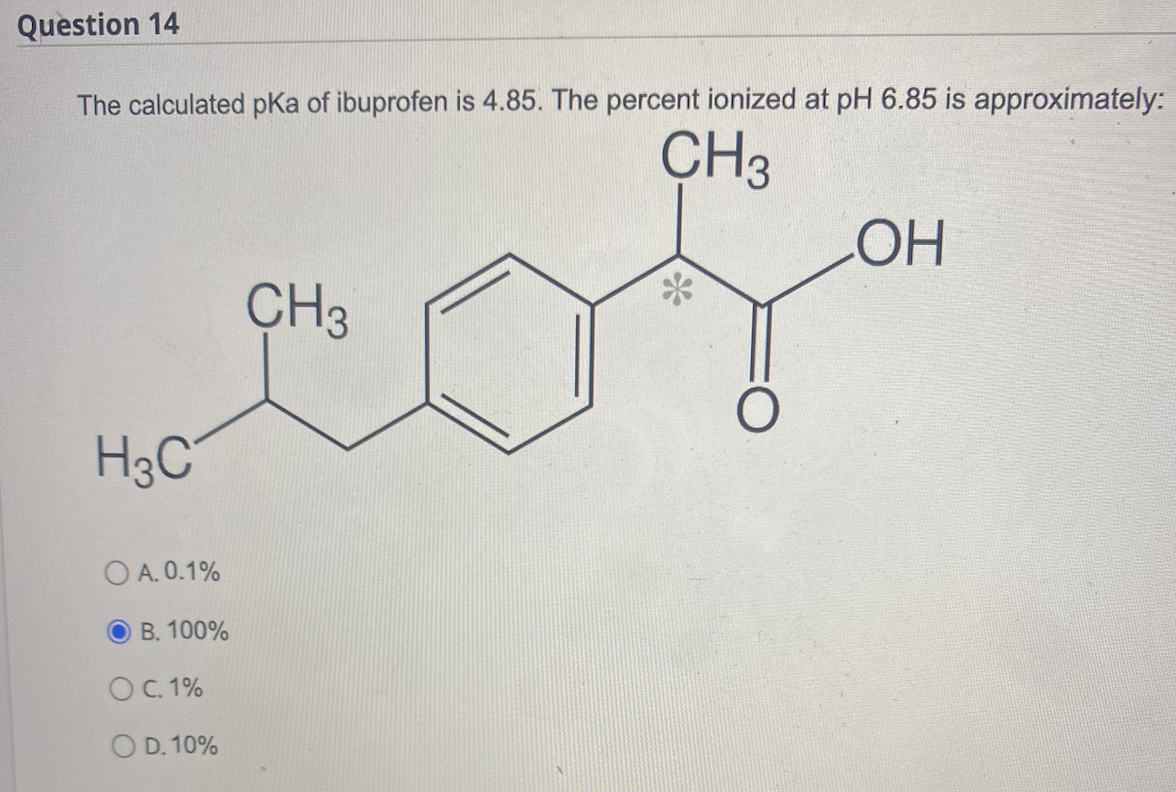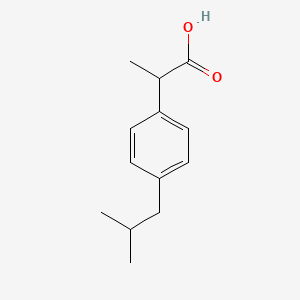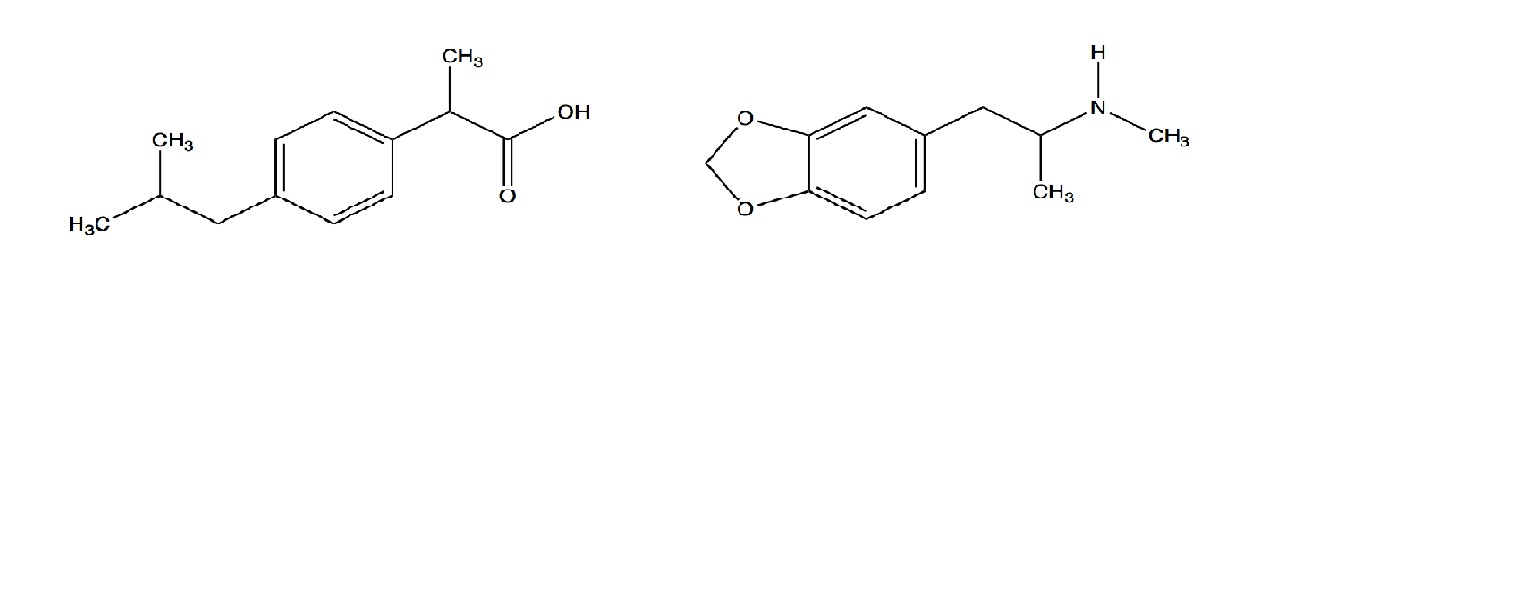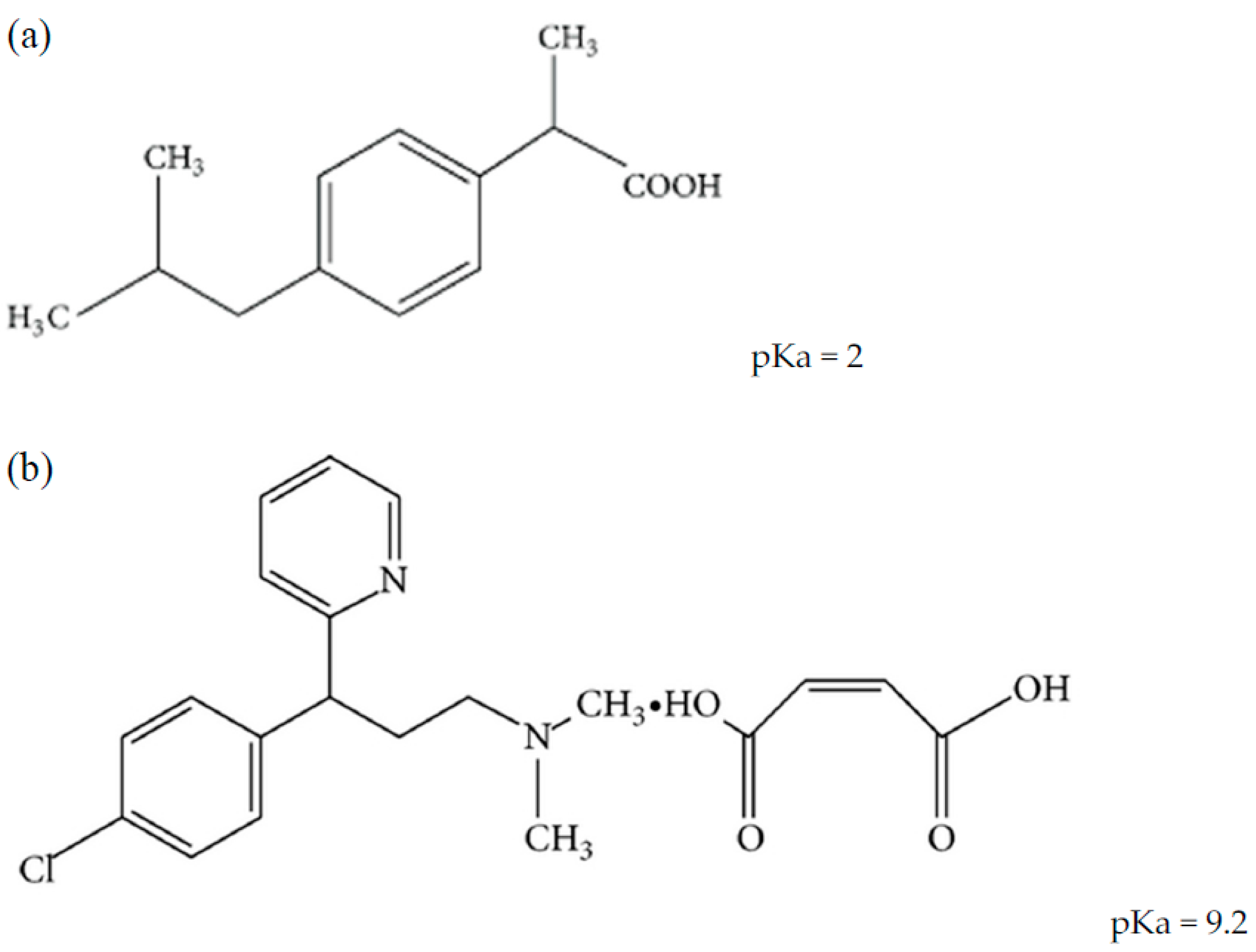
Sci. Pharm. | Free Full-Text | Development of HPLC Method for Simultaneous Determination of Ibuprofen and Chlorpheniramine Maleate
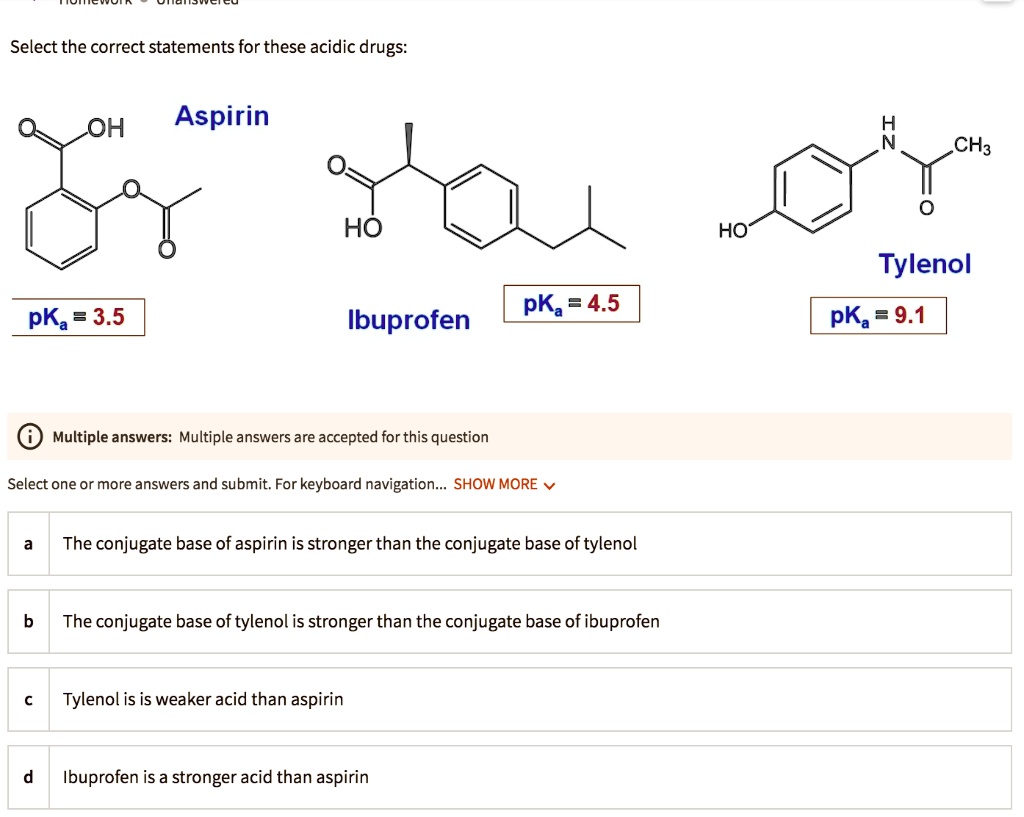
SOLVED: Select the correct statements for these acidic drugs: OH Aspirin HO HO Tylenol pKa 9.1 pKa =4.5 pKa 3.5 Ibuprofen Multiple answers: Multiple answers are accepted for this question Select one
Catalytic Ozonation of Ibuprofen in Aqueous Media over Polyaniline–Derived Nitrogen Containing Carbon Nanostructures

Comparative evaluation of ibuprofen co-crystals prepared by solvent evaporation and hot melt extrusion technology - ScienceDirect
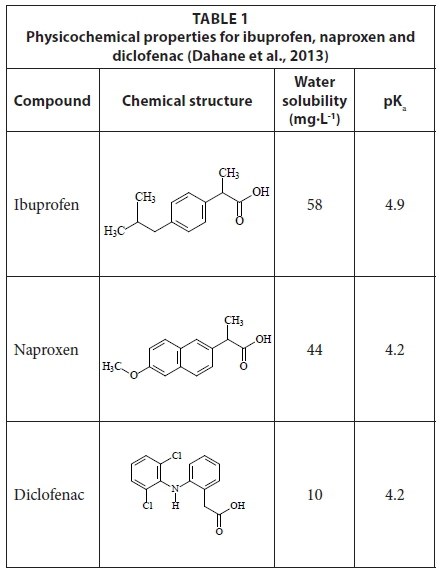
Simultaneous determination of naproxen, ibuprofen and diclofenac in wastewater using solid-phase extraction with high performance liquid chromatography
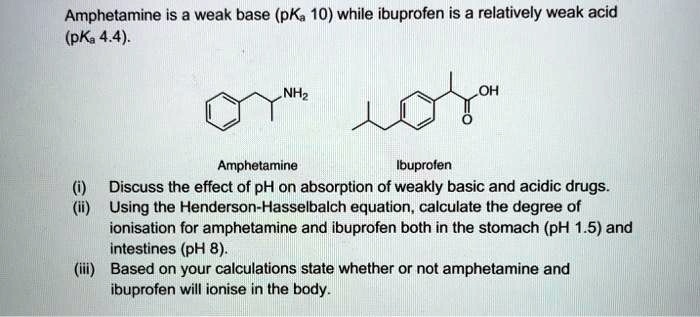
SOLVED: Amphetamine is a weak base (pKa 10) while ibuprofen is a relatively weak acid (pKa 4.4). Discuss the effect of pH on absorption of weakly basic and acidic drugs. Using the

Effect of Water pH on the Uptake of Acidic (Ibuprofen) and Basic (Propranolol) Drugs in a Fish Gill Cell Culture Model | Environmental Science & Technology

Effects of pH, dissolved organic matter, and salinity on ibuprofen sorption on sediment – topic of research paper in Environmental engineering. Download scholarly article PDF and read for free on CyberLeninka open

Ibuprofen: water affinity, effect of acidic pH and resonance structure:... | Download Scientific Diagram

Chemical structures and pKa values of the five different nonsteroidal... | Download Scientific Diagram
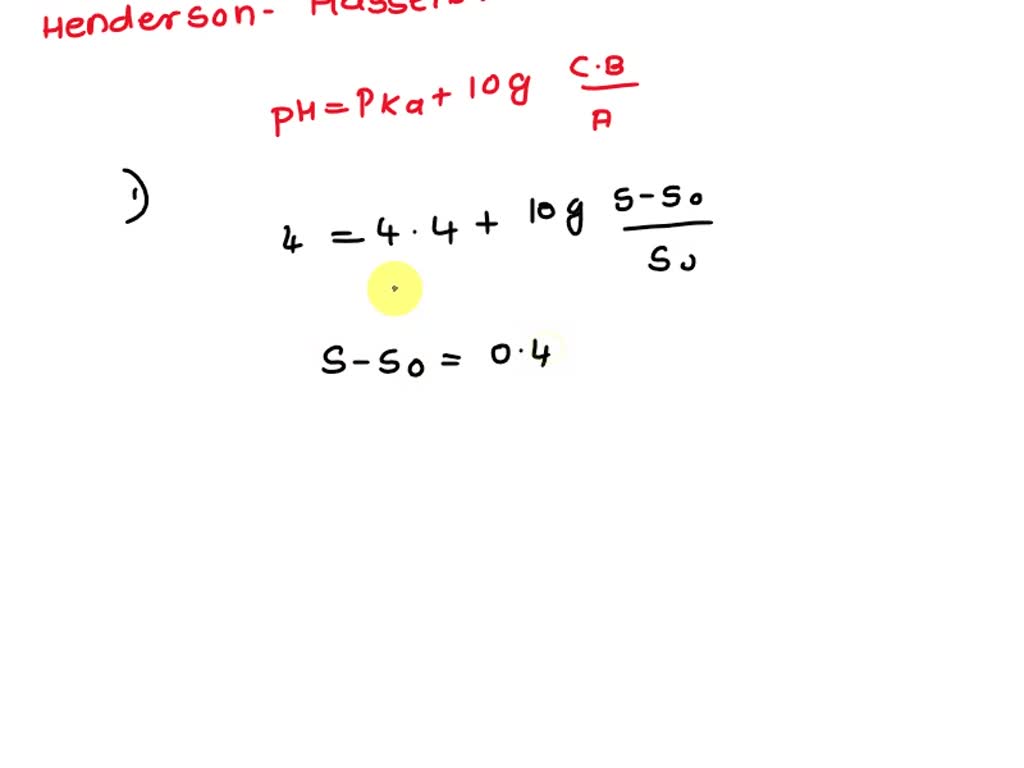
SOLVED: Ibuprofen is an acidic drug with a pKa of 4.4. The solubility of ibuprofen in water at pH 0.5 and 25 C is 5.52 x 10-5 M. At the same temperature,
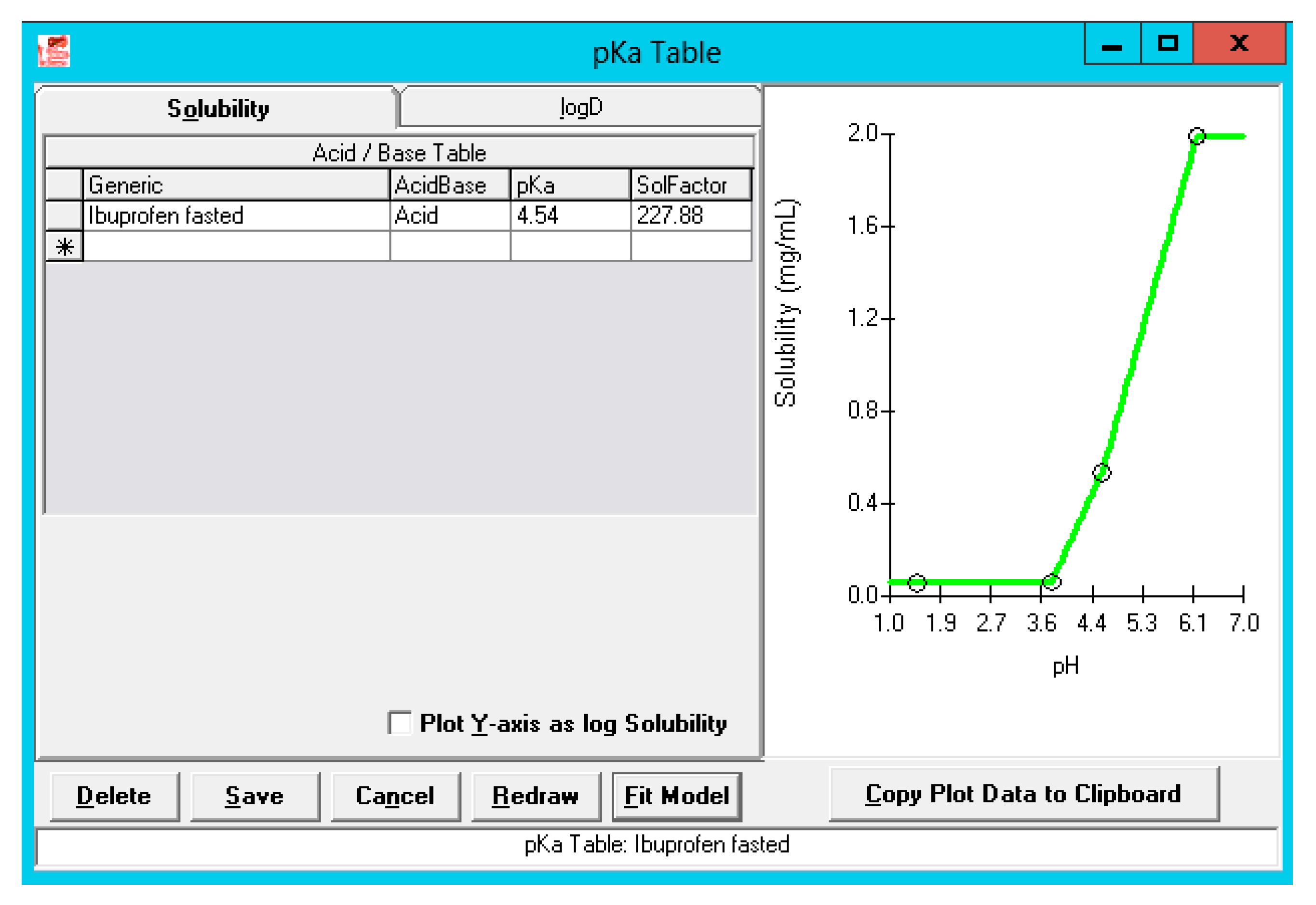
Pharmaceutics | Free Full-Text | A Mechanistic Physiologically-Based Biopharmaceutics Modeling (PBBM) Approach to Assess the In Vivo Performance of an Orally Administered Drug Product: From IVIVC to IVIVP
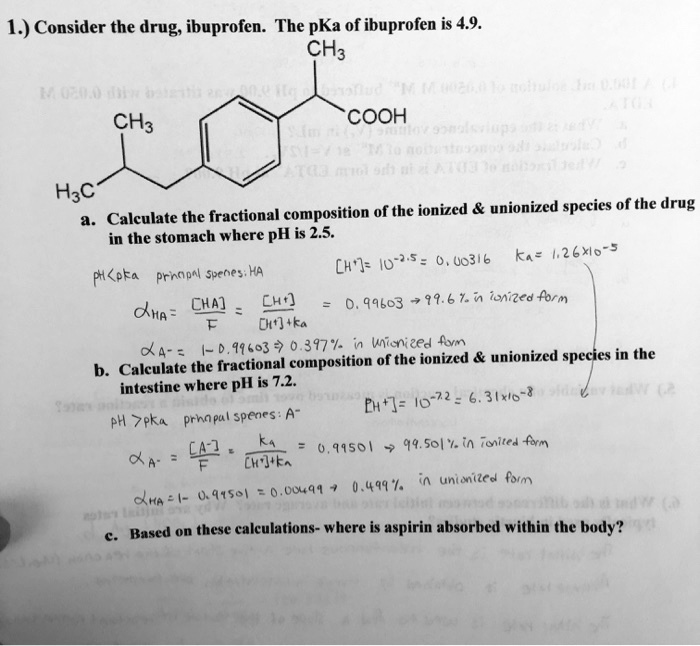
SOLVED: Consider the drug ibuprofen. The pKa of ibuprofen is 4.9. CH3COOH is the fractional composition of the ionized and unionized species of the drug. Calculate in the stomach where pH is